Evaporation and
Intermolecular Attractions
In this experiment, Temperature Probes are placed in various liquids. Evaporation occurs when the probe is removed from the liquid’s container. This evaporation is an endothermic process that results in a temperature decrease. The magnitude of a temperature decrease is, like viscosity and boiling temperature, related to the strength of intermolecular forces of attraction. In this experiment, you will study temperature changes caused by the evaporation of several liquids and relate the temperature changes to the strength of intermolecular forces of attraction. You will use the results to predict, and then measure, the temperature change for several other liquids.
You will encounter two types of organic compounds in this experiment—alkanes and alcohols. The two alkanes are pentane, C5H12, and hexane, C6H14. In addition to carbon and hydrogen atoms, alcohols also contain the -OH functional group. Methanol, CH3OH, and ethanol, C2H5OH, are two of the alcohols that we will use in this experiment. You will examine the molecular structure of alkanes and alcohols for the presence and relative strength of two intermolecular forces—hydrogen bonding and dispersion forces.
OBJECTIVES
In this experiment, you will
• Study temperature changes caused by the evaporation of several liquids.
• Relate the temperature changes to the strength of intermolecular forces of attraction.
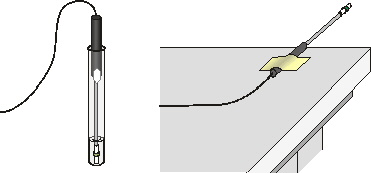
Figure 1
MATERIALS
LabQuest |
methanol (methyl alcohol) |
LabQuest App |
ethanol (ethyl alcohol) |
2 Temperature Probes |
1-propanol |
6 pieces of filter paper (2.5 cm X 2.5 cm) |
1-butanol |
2 small rubber bands |
n-pentane |
masking tape |
n-hexane |
Pre-lab exercise
Prior to doing the experiment, complete the Pre-Lab table. The name and formula are given for each compound. Draw a structural formula for a molecule of each compound. Then determine the molecular weight of each of the molecules. Dispersion forces exist between any two molecules, and generally increase as the molecular weight of the molecule increases. Next, examine each molecule for the presence of hydrogen bonding. Before hydrogen bonding can occur, a hydrogen atom must be bonded directly to an N, O, or F atom within the molecule. Tell whether or not each molecule has hydrogen-bonding capability.
PROCEDURE
1. Obtain and wear goggles! CAUTION: The compounds used in this experiment are flammable and poisonous. Avoid inhaling their vapors. Avoid contacting them with your skin or clothing. Be sure there are no open flames in the lab during this experiment. Notify your teacher immediately if an accident occurs.
2. Connect the Temperature Probes to LabQuest and choose New from the File menu. If you have older sensors that do not auto-ID, manually set up the sensors.
3. On the Meter screen, tap Length. Change the data-collection length to 240 seconds. Select OK. Data collection will last 4 minutes.
4. Wrap Probe 1 and Probe 2 with square pieces of filter paper secured by small rubber bands as shown in Figure 1. Roll the filter paper around the probe tip in the shape of a cylinder. Hint: First slip the rubber band on the probe, wrap the paper around the probe, and then finally slip the rubber band over the paper. The paper should be even with the probe end.
5. Stand Probe 1 in the ethanol container and Probe 2 in the 1-propanol container. Make sure the containers do not tip over.
6. Prepare 2 pieces of masking tape, each about 10 cm long, to be used to tape the probes in position during Step 7.
7. After the probes have been in the liquids for at least 30 seconds, start data collection. A live graph of temperature vs. time for both Probe 1 and Probe 2 is being plotted on the screen. Live readings are displayed to the right of the graph. Monitor the temperature for 15 seconds to establish the initial temperature of each liquid. Then simultaneously remove the probes from the liquids and tape them so the probe tips extend 5 cm over the edge of the table top as shown in Figure 1.
8. Data collection will stop after 4 minutes (or stop data collection before 4 minutes has elapsed). Examine the graph of temperature vs. time. Based on your data, determine the maximum temperature, t1, and minimum temperature, t2 for both probes. Record t1 and t2 for each probe.
9. For each liquid, subtract the minimum temperature from the maximum temperature to determine Δt, the temperature change during evaporation.
10. Based on the Δt values you obtained for these two substances, plus information in the Pre-Lab exercise, predict the size of the Δt value for 1-butanol. Compare its hydrogen-bonding capability and molecular weight to those of ethanol and 1-propanol. Record your predicted Δt, then explain how you arrived at this answer in the space provided. Do the same for n-pentane. It is not important that you predict the exact Δt value; simply estimate a logical value that is higher, lower, or between the previous Δt values.
11. Test your prediction in Step 10 by repeating Steps 5–9 using 1-butanol with Probe 1 and n-pentane with Probe 2.
12. Based on the Δt values you have obtained for all four substances, plus information in the Pre-Lab exercise, predict the Δt values for methanol and n-hexane. Compare the hydrogen-bonding capability and molecular weight of methanol and n-hexane to those of the previous four liquids. Record your predicted Δt, then explain how you arrived at this answer in the space provided.
13. Test your prediction in Step 12 by repeating Steps 5–9, using methanol with Probe 1 and n-hexane with Probe 2.
Processing the data
1. Two of the liquids, n-pentane and 1-butanol, had nearly the same molecular weights, but significantly different Δt values. Explain the difference in Δt values of these substances, based on their intermolecular forces.
2. Which of the alcohols studied has the strongest intermolecular forces of attraction? The weakest intermolecular forces? Explain using the results of this experiment.
3. Which of the alkanes studied has the stronger intermolecular forces of attraction? The weaker intermolecular forces? Explain using the results of this experiment.
4. Plot a graph of Δt values of the four alcohols versus their respective molecular weights. Plot molecular weight on the horizontal axis and Δt on the vertical axis.
PRE-LAB
Substance |
Formula |
Structural Formulas |
Molecular Weight |
Hydrogen Bond |
ethanol |
C2H5OH |
|
|
|
1-propanol |
C3H7OH |
|
|
|
1-butanol |
C4H9OH |
|
|
|
n-pentane |
C5H12 |
|
|
|
methanol |
CH3OH |
|
|
|
n-hexane |
C6H14 |
|
|
|
data table
Substance |
t1
|
t2 |
Δt
(t1– t2) |
|
|
|
ethanol |
|
|
|
|
|
|
1-propanol |
|
|
|
|
Predicted |
Explanation |
1-butanol |
|
|
|
|
|
|
n-pentane |
|
|
|
|
|
|
methanol |
|
|
|
|
|
|
n-hexane |
|
|
|
|
|
|